Search Results for 'high-throughput screening'
-
New project to advance platelet-based therapeutics for patients
CCRM and GE Healthcare Life Sciences develop proprietary media formulations for Platelet BioGenesis
-

What Are the Benefits of an Automated High-Throughput Screening Process for Custom Cell Culture Media Development
We are often asked if culture media development can be customized at the discovery stage of the product, and if feasible, how it can be done.
-
Analytical Services | Bio News | Drug Discovery | Industry News | Laboratory Testing
SGS Implements PBMC Analysis
For the collection & use of peripheral blood mononuclear cells (PBMCs) for early phase clinical trials at its CPU in Belgium09.05.18
-
Breaking News | Collaborations & Alliances | Drug Discovery | Industry News

AMRI, Metrion Enter Drug Discovery Collaboration
Agreement enables customers to access a broader range of drug discovery expertise09.05.18
-
Bio News | cGMP Manufacture | Collaborations & Alliances | Drug Development | Industry News

Codexis, Porton Enter Collaboration
To accelerate the creation and commercialization of new, low-cost, sustainable manufacturing processes04.23.18
-
Breaking News | CRO News | Drug Discovery | Facilities

Charles River Invests in High-Throughput Screening Infrastructure
Signs collaboration agreement with AstraZeneca02.06.18
-
Drug Development | Drug Discovery | Preclinical Outsourcing

Outsourcing Early Drug Development
Birgit Girshick of Charles River discusses trends in the early discovery and development spaceOnline Exclusives01.17.18
-
APIs
Biocatalysis for APIs
Engineered enzymes pave easier, faster road to scalable API manufacturingMichael Aldridge, Codexis 10.11.17
-
APIs
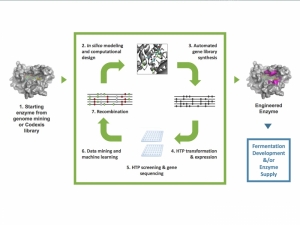
Biocatalysis for APIs
Engineered enzymes pave easier, faster road to scalable API manufacturingMichael Aldridge, SVP of Corporate and Strategic Development, Codexis 09.12.17
-

-
Biologics, Proteins, Vaccines | Drug Development | Drug Discovery | Industry News
Morphotek Launches ADC Services Business
Aligns antibody engineering and clinical development assets into a client-focused ADC business04.03.17
-
Analytical Services | Breaking News | Collaborations & Alliances | Drug Development | Drug Discovery | Industry News
Alcami Expands Analytical Capabilities
Invests in Bruker's D8 Discover HTS System for high-throughput X-Ray diffraction screening03.06.17
-
Drug Discovery | Industry News
Lilly, Regen BioPharma in Drug Discovery Pact
Regen will examine 21,000 Lilly compounds in its NR2F6 high-throughput screening program06.30.16
-
Chemistry | Drug Development | Drug Discovery | Industry News
-
Chemistry | Collaborations & Alliances | Drug Development | R&D
Evotec Reaches Key Padlock Milestone
Achieves key PAD inhibitor preclinical milestone in autoimmune diseases03.10.16
-
Chemistry | Collaborations & Alliances | Drug Discovery
AMRI, NYCNMR in Nanotech Services Alliance
Will co-market nanoscale, formulation and discovery services01.14.16
-
Analytical Services | Chemistry | Collaborations & Alliances | Drug Discovery | Preclinical Outsourcing
AMRI, Multispan in Drug Discovery Services Pact
Alliance aims to increase success in GPCR drug discovery12.14.15
-
Analytical Services | Chemistry | Drug Development | Drug Discovery | Industry News | Laboratory Testing
AMRI Launches Integrated Drug Discovery Center
Aims to increase translation to the clinic leveraging biology, HTS, medicinal chemistry and pharmacology within a single site12.07.15
-












