Search Results for 'single-use systems'
-
Biologics, Proteins, Vaccines

Changes are on the Horizon for Single-use Systems in Pharmaceutical Manufacturing
Single-use systems are cost-effective and produce high-quality pharmaceuticals, but they come with challenges inherent in using plastic materials.Sandi Schaible, Senior Director of Analytical Chemistry, WuXi AppTec 06.07.22
-
Biologics, Proteins, Vaccines
The Evolution of the ADC Manufacturer
How CMDOs can rise to the occasionLisa McDermott and Jyothi Swamy, MilliporeSigma 04.01.20
-

Manufacturing Equipment Technology
The future of highly compliant, efficient equipment that’s customized to suit a large variety of applications.Kristin Brooks, Managing Editor, Contract Pharma 03.04.20
-
Breaking News | Facilities | Industry News

Parker Bioscience Opens Doors to UK Site
Investment includes extended laboratories and new clean room, manufacturing and testing facilities08.16.19
-
Biologics, Proteins, Vaccines
The Biopharma Future
A look at current trends driving volume reductions in biopharmaceutical manufacturing and keys for successful outsourcing partnerships.Karl Pinto and Dave Cunningham, Goodwin Biotechnology 05.07.19
-
cGMP Manufacture

Next-Gen Bioprocessing Manufacturing
Leveraging advances in bioprocessing technology and overcoming the challenges through outsourcingOnline Exclusives Krisitn Brooks, Contract Pharma 04.05.19
-
Breaking News | Collaborations & Alliances

Pall, Broadley-James Partner on SU Technologies
To integrate Pall’s SU technologies with Broadley-James' SU probe and flow sensors for applications across upstream and downstream unit ops03.11.19
-
Drug Development

Future Trends in the CDMO Space
And how to navigate them for successful drug developmentGuillaume Plane, Global Development and Marketing Manager, BioReliance End-to-End Solutions, MilliporeSigma 01.25.19
-
Breaking News | cGMP Manufacture | Industry News

Emergent Expands Commercial Ops Capabilities
Adds ABEC CSR dual purpose single-use systems for large scale 750L microbial fermentation and 500L mammalian cell culture09.28.18
-
Biologics, Proteins, Vaccines | Biosimilars | Drug Development

Haste Makes Waste
When to sprint and when to breathe in the race from lab to clinic.Christian Cattaruzza, MilliporeSigma 09.11.18
-
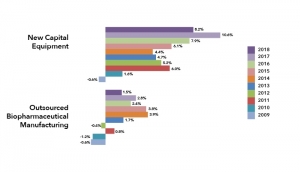
Biopharmaceuticals Outsourcing Slows
Manufacturers are shifting focusEric S. Langer, BioPlan Associates 09.11.18
-
Bio News | Bioanalytical Services | Breaking News | Facilities | Industry News

Saint-Gobain Opens Life Sciences Lab in Mass.
New lab is dedicated to application testing of single-use systems for the cell and gene therapy market05.14.18
-
Biologics, Proteins, Vaccines | Biosimilars

Biopharma Manufacturing Markets
Biomanufacturing contract services and supplies markets continue to expandRonald A. Rader and Eric S. Langer, BioPlan Associates 05.08.18
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Facilities | Industry News

Aldevron Begins Construction of Cleanrooms in $30M GMP Plasmid Facility
AES Clean Technology to design modular cleanrooms for the $30 million, 70,000 sq. ft. manufacturing facility03.23.18
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Industry News

Sartorius Supports ABL Europe Capacity Expansion
Successfully completes Viral Vector GMP manufacturing installation at Strasbourg facility03.19.18
-

Grand River Expands Syringe Filling Capabilities
Adds the Colanar FSV syringe filler at Grand Rapids site03.12.18
-
cGMP Manufacture

Automation in Pharmaceutical Manufacturing
Technological advancements open the door to fundamental performance improvements in pharmaceutical manufacturing.Trevor Marshall, Director of Enterprise System Integration, Zenith Technologies 03.09.18
-
APIs | Aseptic Processing | Breaking News | cGMP Manufacture | Facilities | Fill/Finish | Industry News | Injectables | Packaging & Tracking

VIDEO: Grand River Expands Syringe Filling Capabilities
The Colanar FSV syringe filler will support both small molecule APIs and biologics projects03.06.18
-

2018 Pharma Industry Outlook
The importance of quality and safety, especially in times of changeJohannes Rauschnabel, Chief Pharma Expert, Bosch Packaging 01.30.18
-
cGMP Manufacture | Drug Development | Regulatory Affairs

Contracting & Outsourcing Session Highlights and Key Takeaways
Insight into CMO Trends, Biotrends, Manufacturing Compliance, FDA Quality Agreements, and CMO OversightOnline Exclusives Kristin Brooks, Contract Pharma 10.09.17
-
Biologics, Proteins, Vaccines | cGMP Manufacture

Sartorius Stedim Biotech Introduces New Bioreactor Range
BIOSTAT STR single-use systems aim to reduce bioprocess development timelines and costs08.17.17
-
Bioanalytical Services | Biosimilars
The Analytical Challenges of the Biosimilar Boom
The growing biosimilar industry and its implications on the current drug marketMario DiPaola, Senior Scientific Director, Charles River Laboratories 07.03.17
-
Biologics, Proteins, Vaccines

Trends in New Technologies & Budgets
Budget increases in biopharmaceutical industry indicate future growthEric S. Langer, BioPlan Associates 06.06.17
-
Bioanalytical Services

Current Trends in Analytical Testing
Improved analytical methods continues to be a current need for nearly every aspect of biopharma manufacturingEric S. Langer, BioPlan Associates 05.09.17
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Industry News

Avid Bioservices Expands Manufacturing Capacity
To install multiple Mobius 2000-liter single-use bioreactors05.01.17
-
Biosimilars
CMOs & Biosimilars Mfg. in the U.S.
Biosimilars are moving from novel opportunity to mainstream productEric S. Langer and Ronald A. Rader, BioPlan Associates 04.20.17
-
Bio News | Breaking News | Industry News

Parker domnick hunter Expands European Ops
Will manufacture automated bioprocessing systems at Birtley, UK facility03.29.17
-
Breaking News | Industry News
VWR Acquires JM Separations
Acquisition extends VWR's custom single-use capabilities in Europe06.03.16
-
Analytical Services | Drug Development | Industry News | Laboratory Testing
VWR Acquires JM Separations
Gains single-use fluid handling and separation technology products06.02.16
-
Biosimilars | Clinical Trials | Process Development

Biosimilars Outsourcing and Capacity
CMOs to play important but uncertain role in biosimilars manufacturingRonald A. Rader and Eric S. Langer, BioPlan Associates 05.05.16
-
APIs | Aseptic Processing | Biosimilars | Fill/Finish | Process Development | Supply Chain

Single-use Technology Integral to Advancing Biomanufacturing
Innovative process technologies are a necessity for today’s biopharmaceutical contract manufacturersNigel Walker, Thatu2019s Nice LLC / Nice Insight 03.09.16
-
Breaking News | Extractables and Leachables | Industry News
Chemic Laboratories and Chemical Solutions Join Forces
Collaboration to focus on a variety of pharma and biopharma client interests02.15.16
-
Industry News | Process Validation | Risk Management | Validation
Grand River Expands Disposable Technology Capabilities
Single-use systems aim to offer cost and time savings, improved safety and quality, and flexibility in process design12.09.15
-
Analytical Services | Chemistry | Collaborations & Alliances | Laboratory Testing
Chemic, Gateway Analytical in Joint Analytics Collaboration
Will leverage their collective analytics expertise for the advancement of SUS11.19.15
-
Biosimilars | Process Development
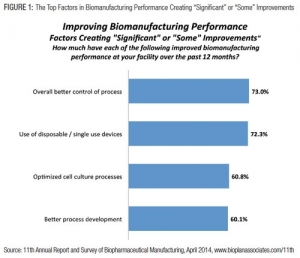
Biopharm Executives Point to Better CMO Performance
Better process control and increased adoption of single-use systems played a part in improving downstream manufacturingEric S. Langer, BioPlan Associates, Inc. 10.14.14
-

CRB’s Marc Pelletier on SIngle-Use Systems at Interphex 2014
More discussion of single-use bioprocess equipment at Interphex.Videos
-
Aseptic Processing | Biologics, Proteins, Vaccines | Extractables and Leachables | Fill/Finish | GMPs/GCPs | Inspections | Laboratory Testing | Microbiology | Parenterals | Regulatory Affairs | Toxicology

Managing E/L in Single-Use Systems
Regulatory and scientific directions in assessing health risksThomas E. Stone 04.05.11












